PQ.Įlectronic Signature, Electronic Records. Quality System requirements to maintain compliant Validations. MDR General Safety requirements.Ĭurrent Good Manufacturing Practices. New European Medical Device Regulations (MDR’s). The FDA approach to Medical Device Classification.ĮU Medical Device Regulation and Classification (per MDD’s). Outline of the FDA regulatory requirements.įDA Medical Device Classification. Current best practices.įDA Medical Device Regulation.

#ISO 13485 TEST METHOD VALIDATION ISO#
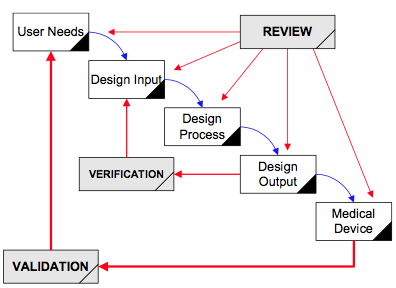
The scope of the ISO 13485 2016 standard includes those organizations which may influence the “life-cycle” of the process under certification, there is also a need to consider processes which have been outsourced. Controls in these areas need to consider activities within supplier organizations.

The introduction within the new revision, specifically includes the addition of storage, distribution, final decommissioning, disposal, and provision for associated activities. What are the changes being introduced under the ISO 13485 2016 revision? There is a 3 year transition period up until 1 st March 2019, during which time, organizations can be certified to either version of the standard, however, after 1 st March 2019, only certification to the ISO 13485 2016 revision will be acceptable, any organization at that time still only complying to the older 2003 revision will not be seen as “certified” to ISO 13485. This revision will now replace the previous ISO 13485 2003 version. Explanation of changes from the 2003 revision.The revised and updated version of the ISO 13485 standard was published on 1 st March 2016.
